
Content
- 1 The Monomer of Polyamide 6: A Direct Answer
- 2 What Is ε-Caprolactam and Where Does It Come From
- 3 How ε-Caprolactam Converts Into Polyamide 6: Ring-Opening Polymerization
- 4 Molecular Structure of Polyamide 6 and What It Means in Practice
- 5 Polyamide 6 vs. Polyamide 6,6: Same Atoms, Different Origins
- 6 Global Production Scale and Commercial Significance of the Caprolactam–PA6 Chain
- 7 Residual Monomer in Polyamide 6: Why It Matters and How It Is Removed
- 8 Key Properties of Polyamide 6 Derived from Its Caprolactam Monomer Structure
- 9 Recycling and Sustainability of Polyamide 6: The Monomer as the End Point
- 10 Applications of Polyamide 6 Across Industries
The Monomer of Polyamide 6: A Direct Answer
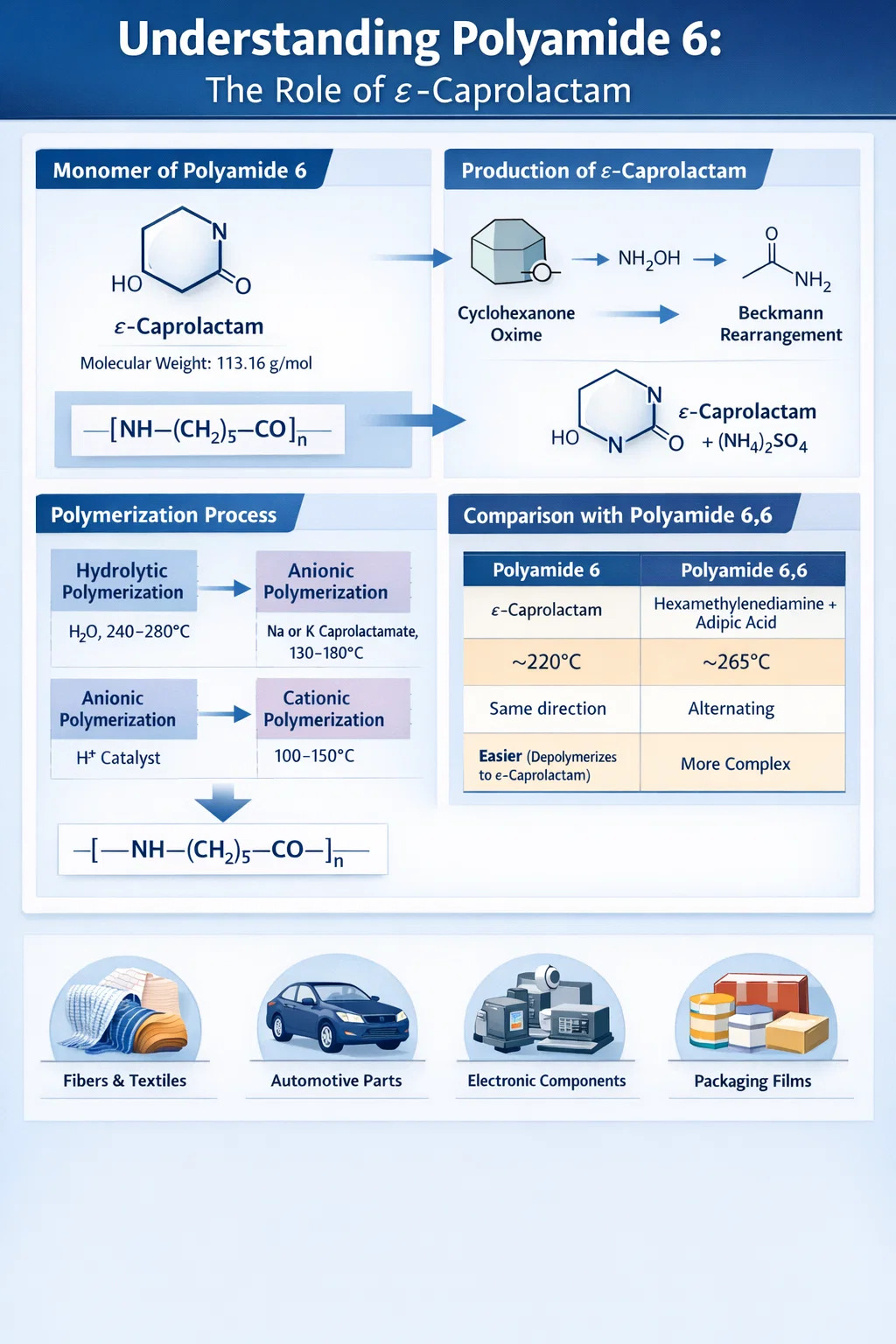
The monomer of polyamide 6 is ε-caprolactam, a cyclic amide (lactam) with the molecular formula C₆H₁₁NO and a molecular weight of 113.16 g/mol. Unlike most condensation polymers that require two distinct monomers, polyamide 6 is produced through the ring-opening polymerization of this single cyclic compound. When ε-caprolactam undergoes ring opening, each monomer unit contributes one amide linkage (–CO–NH–) and five methylene groups (–CH₂–) to the growing polymer chain, resulting in a repeating unit of –[NH–(CH₂)₅–CO]–.
This is a key distinction that separates polyamide 6 from its close structural relative, polyamide 6,6 (nylon 6,6), which is made from two different monomers — hexamethylenediamine and adipic acid. Despite their similar repeating unit chemistry, the two polymers have measurably different thermal and mechanical properties because of how the amide bonds are oriented along the chain. Polyamide 6 has all amide bonds pointing in the same direction, while polyamide 6,6 has alternating orientations.
Understanding the monomer is not just an academic exercise. The choice of ε-caprolactam as the starting material directly controls the polymerization kinetics, the molecular weight distribution of the final polymer, residual monomer content (which can be as high as 8–10% after initial polymerization), and the downstream processing behavior of polyamide 6 in industrial applications.
What Is ε-Caprolactam and Where Does It Come From
ε-Caprolactam is a white crystalline solid at room temperature, with a melting point of approximately 69–71°C and a boiling point near 270°C. It is highly soluble in water and many organic solvents, which is relevant to the aqueous polymerization processes sometimes used in industrial settings. The compound belongs to the class of lactams — cyclic amides formed by intramolecular condensation of an amino acid. Specifically, ε-caprolactam is the cyclic form of 6-aminohexanoic acid (also called ε-aminocaproic acid).
Industrially, ε-caprolactam is produced almost exclusively from cyclohexanone via the Beckmann rearrangement. The process involves three main steps:
- Cyclohexanone reacts with hydroxylamine sulfate to form cyclohexanone oxime.
- The oxime undergoes Beckmann rearrangement in the presence of fuming sulfuric acid (oleum), converting the C=N bond into the amide ring.
- The resulting caprolactam–sulfuric acid mixture is neutralized with ammonia, releasing crude ε-caprolactam and ammonium sulfate as a co-product.
A significant drawback of the traditional route is that it generates roughly 1.6–2 kg of ammonium sulfate per kilogram of caprolactam, creating a large waste stream. This has driven research into alternative synthesis routes, including the gas-phase Beckmann rearrangement over zeolite catalysts, which produces water as the only by-product and has been commercialized by Sumitomo Chemical in Japan since the early 2000s.
The purity of ε-caprolactam used in polymerization must be extremely high — typically above 99.9% — because even trace impurities can act as chain terminators or introduce color into the final polymer. Common impurities that must be carefully controlled include cyclohexanone, cyclohexanone oxime, and various ring-opened amino acid derivatives.
How ε-Caprolactam Converts Into Polyamide 6: Ring-Opening Polymerization
The polymerization of ε-caprolactam to form polyamide 6 occurs through ring-opening polymerization (ROP), which can proceed by three distinct mechanisms depending on the initiator and conditions used. In all cases, the seven-membered ring of caprolactam is opened and the monomer adds to a growing chain.
Hydrolytic Polymerization
This is by far the most widely used industrial route, accounting for the vast majority of global polyamide 6 production. Water (typically 0.5–2% by weight of the monomer charge) acts as the initiator. The process occurs in three overlapping stages:
- Water hydrolyzes caprolactam to 6-aminohexanoic acid, opening the ring.
- The amino acid undergoes polycondensation, forming oligomers with amino and carboxyl end groups.
- Chain growth continues by addition of caprolactam to the amine chain ends (anionic-like addition) alongside further condensation.
In the continuous VK (Vereinfacht Kontinuierlich, meaning simplified continuous) tube reactor process, caprolactam and water are fed at the top of a tall vertical reactor maintained at 240–280°C. The reaction takes approximately 12–20 hours to reach equilibrium, producing a polymer with a number-average molecular weight (Mₙ) typically in the range of 15,000–30,000 g/mol. The equilibrium melt contains roughly 8–10% unconverted caprolactam and low-molecular-weight oligomers, which must be extracted by hot water washing before the polymer can be used in most applications.
Anionic Polymerization
Anionic ring-opening polymerization uses strong base initiators such as sodium caprolactamate or potassium caprolactamate, combined with an activator (N-acyl caprolactam or isocyanate derivatives). This mechanism is much faster than hydrolytic polymerization — polymerization can reach near-complete conversion in minutes at temperatures between 130–180°C, well below the melting point of polyamide 6 (220°C).
Because the polymerization occurs in the solid or semi-solid state, anionic ROP is used for reaction injection molding (RIM) and casting processes where the monomer is poured directly into a mold and polymerizes in situ. This enables production of large, complex parts without the need for melt processing. The resulting polymer tends to have a higher molecular weight and broader molecular weight distribution compared to hydrolytically produced material, and residual monomer content is typically lower — often below 1%.
Cationic Polymerization
Cationic polymerization of caprolactam is initiated by strong acids such as phosphoric acid or hydrochloric acid. This route is considerably slower and less selective than anionic polymerization and is rarely used commercially for polyamide 6 production. It produces polymers with broader molecular weight distributions and generally inferior mechanical properties, making it mainly of academic interest.
Molecular Structure of Polyamide 6 and What It Means in Practice
Every repeat unit of polyamide 6 comes directly from one molecule of ε-caprolactam. The repeat unit —[NH–(CH₂)₅–CO]— contains one amide group and a five-carbon aliphatic chain. This simple structure gives rise to several important structural features:
- Hydrogen bonding: The amide N–H and C=O groups form strong inter-chain hydrogen bonds, contributing to the high melting point (~220°C) and good mechanical strength of polyamide 6.
- Crystallinity: Polyamide 6 is semicrystalline, with a degree of crystallinity typically between 30–50% depending on processing conditions. The crystal structure is predominantly the α-form (monoclinic) at equilibrium, with a metastable γ-form that can be induced by rapid quenching.
- Moisture absorption: The amide groups are hydrophilic. Polyamide 6 absorbs 2.5–3.5% moisture at 50% relative humidity and up to 9–10% at saturation. Absorbed water acts as a plasticizer, reducing tensile modulus but improving impact resistance and elongation at break.
- End groups: Industrial polyamide 6 chains are terminated by amine (–NH₂) and carboxyl (–COOH) groups. The ratio of these end groups can be controlled by adding chain terminators such as acetic acid (to cap amine ends) or hexamethylenediamine (to cap acid ends), which is important for controlling molecular weight and melt viscosity.
The glass transition temperature (Tg) of dry polyamide 6 is approximately 50–60°C, dropping to around 0°C when fully moisture-conditioned. This means that the mechanical properties measured on dry-as-molded specimens are significantly higher than those in actual service conditions, a factor that engineers must account for in design calculations.
Polyamide 6 vs. Polyamide 6,6: Same Atoms, Different Origins
One of the most common points of confusion in polymer chemistry is the relationship between polyamide 6 and polyamide 6,6. Their empirical repeat units are nearly identical — both contain six carbons and one amide group per structural unit — but they differ fundamentally in monomer origin and chain microstructure.
| Property | Polyamide 6 | Polyamide 6,6 |
|---|---|---|
| Monomer(s) | ε-Caprolactam (one monomer) | Hexamethylenediamine + Adipic acid (two monomers) |
| Polymerization type | Ring-opening polymerization | Condensation polymerization |
| Melting point | ~220°C | ~265°C |
| Glass transition temperature (dry) | ~50–60°C | ~70–80°C |
| Moisture absorption (saturation) | ~9–10% | ~8–8.5% |
| Amide bond orientation | All same direction | Alternating directions |
| Processing flexibility | Higher (lower melt viscosity) | Lower (narrower processing window) |
| Recyclability | Easier (depolymerizes back to caprolactam) | More complex chemical recycling |
The higher melting point of polyamide 6,6 gives it an advantage in high-temperature under-the-hood automotive applications, while polyamide 6's lower processing temperature and broader melt processing window make it preferable for fiber spinning and thin-wall injection molding. In the global engineering plastics market, both materials hold substantial positions, with polyamide 6 accounting for roughly 35–40% of total nylon consumption by volume.
Global Production Scale and Commercial Significance of the Caprolactam–PA6 Chain
The commercial scale of ε-caprolactam and polyamide 6 production underscores the practical importance of this monomer–polymer relationship. Global caprolactam production capacity exceeded 7 million metric tons per year as of the early 2020s, with the largest producers located in China, which accounts for over 50% of global output. Major international producers include BASF (Germany), Honeywell (USA), DSM/Fibrant (Netherlands), and AdvanSix (USA).
The end-use breakdown of polyamide 6 is approximately:
- ~60–65% fibers and filaments (carpet yarn, industrial textiles, apparel, tire cord)
- ~20–25% engineering plastics (injection-molded and extruded parts)
- ~10–15% films and other specialty applications
Price fluctuations in caprolactam directly propagate to polyamide 6 pricing because the monomer represents roughly 70–80% of the raw material cost in PA6 production. Caprolactam prices are in turn sensitive to benzene prices (the starting material for cyclohexane, which is oxidized to cyclohexanone), energy costs, and sulfuric acid costs. This tight feedstock linkage makes polyamide 6 one of the commodity engineering materials most exposed to petrochemical market cycles.
Residual Monomer in Polyamide 6: Why It Matters and How It Is Removed
Because the hydrolytic polymerization of ε-caprolactam is an equilibrium reaction, the polyamide 6 melt at the reactor outlet always contains unreacted monomer and oligomers — typically 8–10% by weight. This residual caprolactam and cyclic oligomers must be removed before the polymer can be used for most commercial applications, for several important reasons:
- Mechanical properties: Residual monomer and oligomers act as plasticizers and diluents, reducing tensile strength, modulus, and heat distortion temperature. A fully extracted polyamide 6 typically shows tensile modulus values 10–20% higher than unextracted material.
- Mold deposits: In injection molding, volatile caprolactam evaporates during processing and deposits on mold surfaces, causing surface defects and requiring frequent mold cleaning — a significant production cost in high-volume operations.
- Food contact compliance: Regulatory limits for caprolactam migration from PA6 packaging materials are strict. The EU regulation (EU) 10/2011 sets a specific migration limit (SML) of 15 mg/kg food simulant for caprolactam, requiring that food-contact grades be thoroughly extracted.
- Odor and taste: Even small amounts of caprolactam contribute to objectionable odor in final products, particularly problematic in automotive interior components and food packaging.
The standard extraction process involves washing polymer chips or granules with hot water at 90–100°C in countercurrent extractors. The extraction time and water volume required depend on chip size — smaller chips with larger surface-to-volume ratios extract faster. A typical industrial extraction cycle brings residual monomer content down to below 0.5%, with food-contact grades requiring extraction to below 0.1%. The caprolactam-rich extract water is then evaporated and the recovered caprolactam is recycled back to the polymerization reactor, making the overall process highly atom-efficient.
After extraction, the wet chips must be dried before melt processing to remove absorbed water, which would otherwise cause hydrolytic degradation of amide bonds during extrusion or injection molding. Drying is typically carried out in hopper dryers at 80°C under vacuum or dry air circulation to a moisture content below 0.2% for most applications.
Key Properties of Polyamide 6 Derived from Its Caprolactam Monomer Structure
The properties that make polyamide 6 commercially valuable trace directly back to the chemical structure of ε-caprolactam and the way it polymerizes. The following table summarizes the typical property range for unfilled, conditioned polyamide 6 in comparison to its as-molded (dry) state:
| Property | Dry as-molded | Conditioned (50% RH) |
|---|---|---|
| Tensile strength (MPa) | 75–85 | 50–65 |
| Tensile modulus (GPa) | 2.8–3.3 | 1.2–1.8 |
| Elongation at break (%) | 25–50 | 100–200 |
| Notched Charpy impact (kJ/m²) | 4–6 | 10–20 |
| Heat deflection temperature (°C, 1.8 MPa) | 55–65 | ~45 |
| Melting point (°C) | 215–225 | 215–225 |
The dramatic difference between dry and conditioned properties is one of the most important practical considerations when specifying polyamide 6 for structural applications. Engineers who base their designs on dry-as-molded data risk overestimating stiffness and heat resistance in real operating environments. For glass-fiber reinforced polyamide 6 grades — which are by far the most widely used in structural engineering plastic applications — this moisture sensitivity is much reduced because the fibers dominate the load-bearing behavior.
Recycling and Sustainability of Polyamide 6: The Monomer as the End Point
One of the most compelling aspects of polyamide 6 from a circular economy perspective is that it can be chemically recycled back to its original monomer, ε-caprolactam, with high efficiency. This monomer-to-monomer recyclability is a direct consequence of the ring-opening polymerization mechanism — the same equilibrium that produces residual caprolactam in the reactor can be exploited in reverse under depolymerization conditions.
The depolymerization of polyamide 6 to caprolactam is carried out industrially by heating the polymer in superheated steam at temperatures around 250–350°C in the presence of phosphoric acid or other catalysts. Recovery yields of caprolactam can exceed 90% under optimized conditions. The recovered caprolactam is purified and fed back into the polymerization process, creating a true closed-loop recycling system. Companies such as Aquafil (Italy) have commercialized this approach at industrial scale through their ECONYL® process, which processes post-consumer carpet waste, fishing nets, and industrial nylon scraps into virgin-equivalent caprolactam.
Chemical recycling of polyamide 6 is technically more straightforward than that of polyamide 6,6, which requires more complex depolymerization chemistry to break the two different monomers out of the condensation polymer chain. This is considered a meaningful sustainability advantage for polyamide 6 as pressure mounts on the plastics industry to demonstrate viable end-of-life solutions.
Bio-based routes to ε-caprolactam are also under active development, with several research groups working on fermentation routes to lysine and 6-aminohexanoic acid as precursors. While no fully bio-based caprolactam has yet reached commercial-scale production, partial bio-based content through bio-cyclohexanol or bio-adipic acid intermediates has been demonstrated at pilot scale, positioning polyamide 6 for a potential reduction in its fossil feedstock dependency over the coming decade.
Applications of Polyamide 6 Across Industries
The unique combination of properties that flows from the caprolactam-derived structure — strength, toughness, wear resistance, and chemical resistance — positions polyamide 6 across a remarkably wide range of applications.
Fiber and Textile Applications
The largest single use of polyamide 6 is in continuous filament yarn for carpets and rugs, where its excellent wear resistance and resilience — the ability to recover from foot traffic — are critical. Bulk continuous filament (BCF) yarn for carpets accounts for a large share of global PA6 fiber production. Polyamide 6 is also used extensively in apparel (activewear, hosiery, swimwear), where its combination of strength, stretch recovery, and dyeability is valued. Industrial applications include tire cord, conveyor belts, ropes, parachute fabric, and airbag fabric, where high tenacity yarn with specific elongation and energy absorption characteristics is required.
Automotive Components
Glass-fiber reinforced polyamide 6 (typically 30–50% glass fiber by weight) is one of the most widely used materials in automotive plastic components. Key applications include engine covers, air intake manifolds, charge air cooler end caps, oil pans, fan blades, and various structural brackets and housings. The material offers a good balance of stiffness, heat resistance, and dimensional stability that allows metal replacement in semi-structural applications, contributing to vehicle lightweighting. A typical passenger vehicle contains 4–8 kg of polyamide in various components, with PA6 and PA6,6 sharing this market roughly equally.
Electrical and Electronics
Flame-retardant grades of polyamide 6, compounded with halogen-free or halogenated flame retardants to meet UL 94 V-0 ratings, are widely used in connectors, relay housings, circuit breaker components, cable ties, and enclosures. The inherent tracking resistance and mechanical robustness of polyamide 6 make it well suited for components that experience both thermal cycling and mechanical stress during service. With the trend toward higher-temperature under-the-hood electronics and electric vehicle battery systems, there is growing demand for heat-stabilized PA6 grades that maintain performance at continuous service temperatures above 130°C.
Packaging and Films
Biaxially oriented polyamide 6 film (BOPA film) is used extensively in food packaging because of its excellent oxygen barrier properties, puncture resistance, and ability to withstand retort sterilization temperatures. Typical oxygen transmission rates for a 15 µm BOPA film are around 40–60 cc/m²·day·atm, significantly better than oriented polypropylene or polyethylene. BOPA film is commonly used in vacuum-packed meat and cheese packaging, boil-in-bag applications, and as the structural layer in multilayer barrier films.



 English
English 中文简体
中文简体 Español
Español русский
русский عربى
عربى





